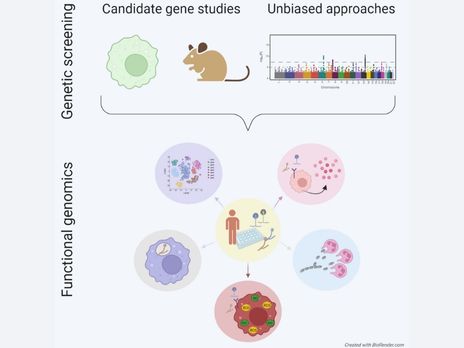
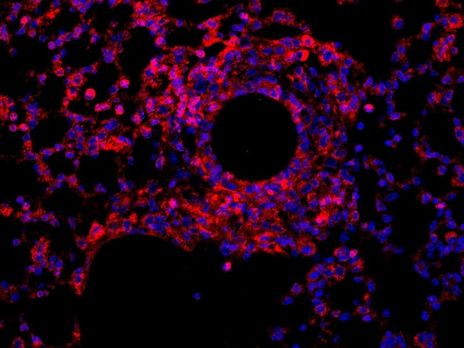
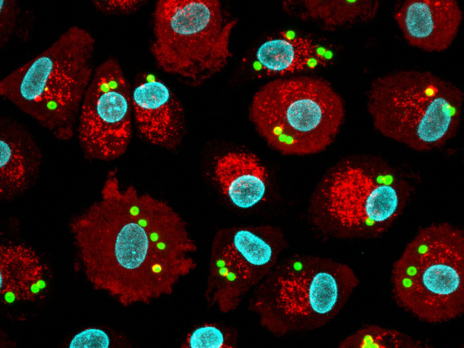
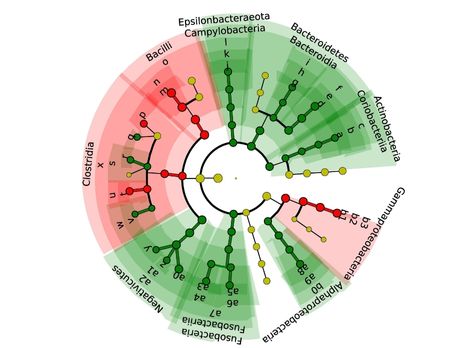
Scientific Highlights
(1) Feys S, Gonçalves SM, Khan M, Choi S, Boeckx B, Chatelain D, Cunha C, Debaveye Y, Hermans G, Hertoghs M, Humblet-Baron S, Jacobs C, Lagrou K, Marcelis L, Maizel J, Meersseman P, Nyga R, Seldeslachts L, Starick MR, Thevissen K, Vandenbriele C, Vanderbeke L, Vande Velde G, Van Regenmortel N, Vanstapel A, Vanmassenhove S, Wilmer A, Van De Veerdonk FL, De Hertogh G, Mombaerts P, Lambrechts D, Carvalho A, Van Weyenbergh J, Wauters J. “Lung epithelial and myeloid innate immunity in influenza-associated or COVID-19-associated pulmonary aspergillosis: an observational study”. Lancet Respir Med. 2022; in press (2) Hoenigl M, Seidel D, Sprute R, Cunha C, Oliverio M, Goldman GH, Ibrahim AS, Carvalho A. “COVID-19-associated fungal infections”. Nat Microbiol. 2022; 7(8):1127-1140; (2) Gonçales RA, Bastos HN, Duarte-Oliveira C, Antunes D, Sokhatska O, Jacob M, Rolo R, Campos CF, Sasaki SD, Donato A, Mapelli SN, Costa S, Souto Moura C, Delgado L, Morais A, Torrado E, van de Veerdonk FL, Weichhart T, Lambris JD, Silvestre R, Garlanda C, Mantovani A, Cunha C, Carvalho A. “Pentraxin 3 inhibits complement-mediated macrophage activation to restrain granuloma formation in sarcoidosis“. Am J Respir Crit Care Med. 2022; epub; (4) Gonçalves SM, Antunes D, Leite L, Mercier T, Horst RT, Vieira J, Espada E, Pinho Vaz C, Branca R, Campilho F, Freitas F, Ligeiro D, Marques A, van de Veerdonk FL, Joosten LAB, Lagrou K, Maertens J, Netea MG, Lacerda JF, Campos A Jr, Cunha C, Carvalho A. “Genetic variation in PFKFB3 impairs antifungal immunometabolic responses and predisposes to invasive pulmonary aspergillosis”. mBio. 2021; 12(3):e0036921; (5) Gonçalves SM, Duarte-Oliveira C, Campos CF, Aimanianda V, Ter Horst R, Leite L, Mercier T, Pereira P, Fernández-García M, Antunes D, Rodrigues CS, Barbosa-Matos C, Gaifem J, Mesquita I, Marques A, Osório NS, Torrado E, Rodrigues F, Costa S, Joosten LA, Lagrou K, Maertens J, Lacerda JF, Campos A Jr, Brown GD, Brakhage AA, Barbas C, Silvestre R, van de Veerdonk FL, Chamilos G, Netea MG, Latgé JP, Cunha C, Carvalho A. “Phagosomal removal of fungal melanin reprograms macrophage metabolism to promote antifungal immunity”. Nat Commun. 2020; 11(1):2282; (6) Kyrmizi I, Ferreira H, Carvalho A, Figueroa JAL, Zarmpas P, Cunha C, Akoumianaki T, Stylianou K, Deepe GS Jr, Samonis G, Lacerda JF, Campos A Jr, Kontoyiannis DP, Mihalopoulos N, Kwon-Chung KJ, El-Benna J, Valsecchi I, Beauvais A, Brakhage AA, Neves NM, Latge JP, Chamilos G. “Calcium sequestration by fungal melanin inhibits calcium-calmodulin signalling to prevent LC3-associated phagocytosis”. Nat Microbiol. 2018; 3(7):791-803; (7) Stappers MHT, Clark AE, Aimanianda V, Bidula S, Reid DM, Asamaphan P, Hardison SE, Dambuza IM, Valsecchi I, Kerscher B, Plato A, Wallace CA, Yuecel R, Hebecker B, da Glória Teixeira Sousa M, Cunha C, Liu Y, Feizi T, Brakhage AA, Kwon-Chung KJ, Gow NAR, Zanda M, Piras M, Zanato C, Jaeger M, Netea MG, van de Veerdonk FL, Lacerda JF, Campos A, Carvalho A, Willment JA, Latgé JP, Brown GD. “Recognition of DHN-melanin by a C-type lectin receptor is required for immunity to Aspergillus”. Nature. 2018; 555(7696):382-386; (8) Cunha C, Gonçalves SM, Duarte-Oliveira C, Leite L, Lagrou K, Marques A, Lupiañez CB, Mesquita I, Gaifem J, Barbosa AM, Pinho Vaz C, Branca R, Campilho F, Freitas F, Ligeiro D, Lass-Flörl C, Löffler J, Jurado M, Saraiva M, Kurzai O, Rodrigues F, Castro AG, Silvestre R, Sainz J, Maertens JA, Torrado E, Jacobsen ID, Lacerda JF, Campos A Jr, Carvalho A. “IL-10 overexpression predisposes to invasive aspergillosis by suppressing antifungal immunity”. J Allergy Clin Immunol. 2017; 140(3):867-870.e9; (9) Cunha C, Aversa F, Lacerda JF, Busca A, Kurzai O, Grube M, Löffler J, Maertens JA, Bell AS, Inforzato A, Barbati E, Almeida B, Santos e Sousa P, Barbui A, Potenza L, Caira M, Rodrigues F, Salvatori G, Pagano L, Luppi M, Mantovani A, Velardi A, Romani L, Carvalho A. “Genetic PTX3 deficiency and aspergillosis in stem-cell transplantation”. N Engl J Med. 2014; 370(5):421-32; (10) Zelante T, Iannitti RG, Cunha C, De Luca A, Giovannini G, Pieraccini G, Zecchi R, D’Angelo C, Massi-Benedetti C, Fallarino F, Carvalho A, Puccetti P, Romani L. “Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22”. Immunity. 2013; 39(2):372-85