Cancer Biomarkers and Therapeutics
Cancer is a leading cause of death worldwide and the global cancer burden is expected to increase in the next decades. Thus, efforts to implement effective preventive measures and better patient care are crucial for cancer control.
Prevention remains the keystone for diminishing cancer mortality, and increasing knowledge on cancer epidemiology and etiology is crucial for identifying adequate preventive measures. Knowing the cancer incidence trends and risk factors are critical for designing strategies to improve prevention and increase early detection, ideally at a curative stage. There have been important advances in cancer treatment over the years, aiming at more effective and safer therapies. However, clinical response to anticancer drugs is not equal in all patients and ultimately there is development of resistance, which poses a major hurdle to patients, and a challenge for physicians and researchers. Another important need is the identification of clinically-relevant cancer biomarkers, that have manifold applications, from risk assessment, to screening, differential diagnosis, prognosis, prediction of treatment response, and monitoring disease progression.
The BiTCancer team comprises researchers at different career stages, with multidisciplinar background and complementary expertise. Over the years, the team members have been working on these cancer needs using up-to-date approaches and models (in vitro, in vivo, in silico, patient samples). The main goals of the team are to uncover i) new cancer risk biomarkers, ii) new biomarkers of diagnosis, prognosis and response to therapy, iii) novel strategies to treat cancer and overcome resistance to classical and targeted therapy, aiming to offer novel personalized therapies to patients.
Team Members


Bruno M. Costa

Rui M. Reis

Adhemar Longatto-Filho

Céline S. Gonçalves

Luís Pacheco-Figueiredo

Ricardo Jorge Fernandes

Sandra F. Martins

Ana Raquel-Cunha

Eduarda P. Martins

Joana Pereira-Vieira

Julieta Afonso

Marta Viana-Pereira

Sara Granja

Adriana Mendes

Ana Silva

Francisco Botelho

Joana M. Ferreira

Joana Pinheiro

Mónica Cerqueira

Patrícia Fontão

Rafaela Oliveira

Sofia Sousa

Sónia Pires Celeiro
Rita Leite

Helena M. Figueiredo
Team Members

Fátima Baltazar
Principal Investigator
Team Coordinator

Bruno M. Costa
Principal Investigator

Rui M. Reis
Principal Investigator

Adhemar Longatto-Filho
Senior Researcher

Céline S. Gonçalves
Assistant Researcher

Marta Costa
Assistant Researcher

Luís Pacheco-Figueiredo
Clinician-Researcher

Ricardo Jorge Fernandes
Clinician-Researcher

Sandra F. Martins
Clinician-Researcher

Julieta Afonso
Postdoctoral Researcher

Olga Martinho
Postdoctoral Researcher

Marta Viana-Pereira
Affiliated Researcher

Rafaela Dias Oliveira
Affiliated Researcher

Sara Granja
Affiliated Researcher

Adriana Mendes
PhD Student

Ana Raquel Bertão
PhD Student

Ana Raquel-Cunha
PhD Student

Ana Silva
PhD Student

Andreia Pereira-Nunes
PhD Student

Diana Cardoso-Carneiro
PhD Student

Eduarda P. Martins
PhD Student

Joana Pinheiro
PhD Student

Joana Pereira-Vieira
PhD Student

Mónica Cerqueira
PhD Student

Patrícia Fontão
PhD Student

Sofia Sousa
PhD Student

Sónia Pires Celeiro
PhD Student

Viktoriya Ivasiv
PhD Student

Joana M. Ferreira
MSc Student

Rafael Azevedo
MSc Student

Rui Marques
MSc Student

Maria Borges
MSc Student
Projects
- Rui M. Reis
- Prostate cancer (PCa) is the second leading cause of cancer-related deaths in men and therapy resistance is a major problem for PCa patients. Recently, we identified…
- Bruno M. Costa
- Of all brain tumors, glioblastoma (GBM) is particularly devastating, being the most common and most malignant type. Using a wide panoply of biological samples, ranging from…
- Bruno M. Costa
- Glioblastoma (GBM) is a notoriously devastating malignant brain tumor, presenting remarkable intratumor heterogeneity. Despite aggressive treatments, it is universally fatal, with a median patient survival of…
- Rui M. Reis
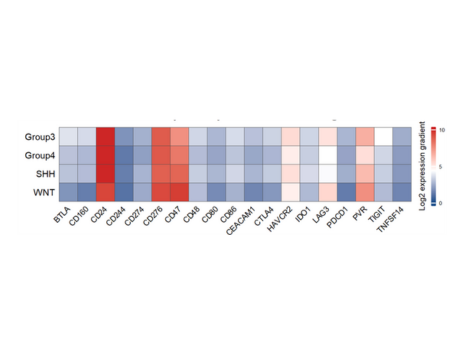
- This project aims to determine the CD24 protein’s biological role and assess its blockade’s impact on the interaction between tumor cells and the immune system…
- Fátima Baltazar
- Reprogramming of energy metabolism is a key hallmark of cancer. Most cancer cells undergo the Warburg effect, with enhanced glycolytic metabolism and lactate production, which provides…
- Bruno M. Costa
- Gliomas, the most common primary brain tumors, arise from the glial cells and typically exhibit bad prognosis. In particular, glioblastoma (GBM), the most common and aggressive…
- Olga Martinho
- Raf kinase inhibitor protein (RKIP), was first described as an inhibitor of MAPK pathway, which accounts for its current name. However, subsequent studies have shown that…
- Fátima Baltazar
- Remote patient monitoring (RPM) of cancer diseases can be potentially used to increase current predictive rates, while contributing for a more cost-effective and accessible diagnosis and treatment…
- Céline S. Gonçalves
- Brain tumors have one of the highest mortality rates and rank 1st in average years of life lost among all tumor types. Among them, glioblastoma (GBM)…
Success Story
The team has vast research history on studying different already established oncogenes that are potential targets for cancer treatment.
TKs (tyrosine kinases) are probably the mostly explored molecules in targeted therapies. We have been studying the response of different cancer types to TK inhibitors already in the clinics, finding new strategies to boost their response (doi: 10.7150/thno.17154).
Examples of new molecular targets are MCTs, Brachyury, RKIP, HOXA9 and WNT proteins that may be targeted in cancer. Monocarboxylate transporters (MCTs) are key molecules in the maintenance of the main metabolic phenotype of cancer cells and are overexpressed in a variety of malignancies. Our studies on MCTs using patient samples and cancer models (doi: 10.1093/neuonc/nos298) contributed to the recognition of MCT1 as target in cancer, which motivated the launching of clinical trials with the MCT1 inhibitor AZD3956 (NCT01791595). In glioblastoma (GBM), we identified HOXA9 as a novel biomarker of poor prognosis in patients, and outlined the critical HOXA9-driven pathways conferring a highly aggressive phenotype and resistance to chemotherapy (doi: 10.18632/oncotarget.3150). We also explored the HOXA9-target genes, of which WNT6 was establishment as a critical oncogene for GBM patients (doi: 10.7150/thno.25025 and International Patent WO/2019/207512A2). Other novel prognostic biomarkers in GBM are Cadherin-3 (doi: 10.1002/1878-0261.13162) and the long non-coding RNA HOTAIR (doi: 10.18632/oncotarget.24597), constituting also novel therapeutic targets. Another key molecule is Brachyury, a crucial development gene that showed to behave as oncogene in different cancer types (doi: 10.1172/JCI38379), leading these studies to further vaccine developments. In GBM, this gene showed a distinct role as tumor suppressor and impacted temozolomide response (doi: 10.1007/s13311-020-00911-9). Many of these biomarkers play a critical role in the crosstalk between malignant and non-malignant cells (e.g. endothelial cells, fibroblasts and immune cells) within the tumour microenvironment.
We have been exploiting these interactions for therapeutic purposes, namely to boost response to novel immunotherapeutic agents (doi.org/10.1038/s41585-019-0263-6). We have also been developing and testing new synthetic molecules for cancer therapy (DOI: 10.1016/j.ejmech.2018.07.058). An example of success is the family of molecules described in the International Patent – WO/2020/261242, that has very promising anticancer activity in poor prognosis cancers, accompanied by a good safety profile, and a novel mechanism of action.
Selected Research Outputs
CHROMENE-BASED COMPOUNDS, METHODS AND USES THEREOF. Costa, Marta; Maria Fernanda R. Paiva Proença; Fátima Baltazar; Patrícia Maciel; Olívia Pontes. 2020. Ref. International Patent - WO/2020/261242.
Afonso J, Santos L, Longatto-Filho A, and Baltazar F. Competitive glucose metabolism in tumour and immune cells – A target to boost bladder cancer immunotherapy? Nat Rev Urol. 2020 Feb;17(2):77-106. Review. doi: 10.1038/s41585-019-0263-6.
Pinto F, Costa ÂM, Santos GC, Matsushita MM, Costa S, Silva VA, Miranda-Gonçalves V, Lopes CM, Clara CA, Becker AP, Neder L, Hajj GN, da Cunha IW, Jones C, Andrade RP, Reis RM. The T-box transcription factor brachyury behaves as a tumor suppressor in gliomas. J Pathol. 2020 May;251(1):87-99. doi: 10.1002/path.5419.
“Singh M, Afonso J, Sharma D, Gupta R, Kumar V, Rani R, Baltazar F*, Kumar V*. Targeting monocarboxylate transporters (MCTs) in cancer: How close are we to the clinics? Semin Cancer Biol. 2023 May;90:1-14. doi: 10.1016/j.semcancer.2023.01.007.”
Céline S. Gonçalves, Nuno Sousa, Bruno M. Costa (2019). A novel oncogene biomarker, methods and uses thereof. International patent no. WO/2019/207512A2.
Miranda-Gonçalves V, Honavar M, Pinheiro C, Martinho O, Cordeiro M, Bebiano G, Costa P, Reis RM, Baltazar F.Monocarboxylate transporters (MCTs) in gliomas: Expression and exploitation as therapeutic targets. Neuro-Oncology, 2013; 15:172-88. doi: 10.1093/neuonc/nos298.
Martinho O, Silva-Oliveira R, Cury FP, Barbosa AM, Granja S, Evangelista AF, Marques F, Miranda-Gonçalves V, Cardoso-Carneiro D, de Paula FE, Zanon M, Scapulatempo-Neto C, Moreira MA, Baltazar F, Longatto-Filho A, Reis RM. HER Family Receptors are Important Theranostic Biomarkers for Cervical Cancer: Blocking Glucose Metabolism Enhances the Therapeutic Effect of HER Inhibitors. Theranostics. 2017 Jan 15;7(3):717-732. doi: 10.7150/thno.17154.
Céline S. Gonçalves, Joana Vieira de Castro, Marta Pojo, Eduarda P. Martins, Sandro Queirós, Emmanuel Chautard, Ricardo Taipa, Manuel Melo Pires, Afonso A. Pinto, Fernando Pardal, Carlos Custódia, Cláudia C. Faria, Carlos Clara, Rui M. Reis, Nuno Sousa, and Bruno M. Costa. WNT6 is a Novel Oncogenic Prognostic Biomarker in Human Glioblastoma, Theranostics, 2018; 8(17): 4805-4823. doi: 10.7150/thno.25025.
Comprehensive Characterization of Cancer Driver Genes and Mutations. Bailey MH, Tokheim C, Porta-Pardo E, Sengupta S, Bertrand D, Weerasinghe A, Colaprico A, Wendl MC, Kim J, Reardon B, Ng PK, Jeong KJ, Cao S, Wang Z, Gao J, Gao Q, Wang F, Liu EM, Mularoni L, Rubio-Perez C, Nagarajan N, Cortés-Ciriano I, Zhou DC, Liang WW, Hess JM, Yellapantula VD, Tamborero D, Gonzalez-Perez A, Suphavilai C, Ko JY, Khurana E, Park PJ, Van Allen EM, Liang H; MC3 Working Group; Cancer Genome Atlas Research Network, Lawrence MS, Godzik A, Lopez-Bigas N, Stuart J, Wheeler D, Getz G, Chen K, Lazar AJ, Mills GB, Karchin R, Ding L. Cell. 2018 Apr 5;173(2):371-385.e18. doi: 10.1016/j.cell.2018.02.06"
Witjes JA, Babjuk M, Bellmunt J, Bruins HM, De Reijke TM, De Santis M, Gillessen S, James N, Maclennan S, Palou J, Powles T, Ribal MJ, Shariat SF, Der Kwast TV, Xylinas E, Agarwal N, Arends T, Bamias A, Birtle A, Black PC, Bochner BH, Bolla M, Boormans JL, Bossi A, Briganti A, Brummelhuis I, Burger M, Castellano D, Cathomas R, Chiti A, Choudhury A, Compérat E, Crabb S, Culine S, De Bari B, De Blok W, J L De Visschere P, Decaestecker K, Dimitropoulos K, Dominguez-Escrig JL, Fanti S, Fonteyne V, Frydenberg M, Futterer JJ, Gakis G, Geavlete B, Gontero P, Grubmüller B, Hafeez S, Hansel DE, Hartmann A, Hayne D, Henry AM, Hernandez V, Herr H, Herrmann K, Hoskin P, Huguet J, Jereczek-Fossa BA, Jones R, Kamat AM, Khoo V, Kiltie AE, Krege S, Ladoire S, Lara PC, Leliveld A, Linares-Espinós E, Løgager V, Lorch A, Loriot Y, Meijer R, Mir MC, Moschini M, Mostafid H, Müller AC, Müller CR, N'Dow J, Necchi A, Neuzillet Y, Oddens JR, Oldenburg J, Osanto S, J G Oyen W, Pacheco-Figueiredo L, Pappot H, Patel MI, Pieters BR, Plass K, Remzi M, Retz M, Richenberg J, Rink M, Roghmann F, Rosenberg JE, Rouprêt M, Rouvière O, Salembier C, Salminen A, Sargos P, Sengupta S, Sherif A, Smeenk RJ, Smits A, Stenzl A, Thalmann GN, Tombal B, Turkbey B, Lauridsen SV, Valdagni R, Van Der Heijden AG, Van Poppel H, Vartolomei MD, Veskimäe E, Vilaseca A, Rivera FAV, Wiegel T, Wiklund P, Williams A, Zigeuner R, Horwich A. EAU-ESMO Consensus Statements on the Management of Advanced and Variant Bladder Cancer-An International Collaborative Multistakeholder Effort†: Under the Auspices of the EAU-ESMO Guidelines Committees. Eur Urol. 2020 Feb;77(2):223-250. doi: 10.1016/j.eururo.2019.09.035.
Andrew Mancini, Ana Xavier-Magalhães, Wendy S Woods, Kien-Thiet Nguyen, Alexandra M Amen, Josie L Hayes, Christof Fellmann, Michael Gapinske, Andrew M McKinney, Chibo Hong, Lindsey E Jones, Kyle M Walsh, Robert J A Bell, Jennifer A Doudna, Bruno M Costa, Jun S Song, Pablo Perez-Pinera, Joseph F Costello. Disruption of the β1L Isoform of GABP Reverses Glioblastoma Replicative Immortality in a TERT Promoter Mutation-Dependent Manner. Cancer Cell. 2018 Sep 10;34(3):513-528.e8. doi: 10.1016/j.ccell.2018.08.003.



Contact us
Phone: +351 253 604 967
Fax: +351 253 604 809
Email: icvs.sec@med.uminho.pt
Address
Life and Health Sciences
Research Institute (ICVS)
School of Medicine,
University of Minho,
Campus de Gualtar
4710-057 Braga
Portugal

Copyright ©2026 ICVS. All Rights Reserved. Developed by TCIT



Copyright ©2026 ICVS. All Rights Reserved. Developed by TCIT
Address
Life and Health Sciences
Research Institute (ICVS)
School of Medicine,
University of Minho,
Campus de Gualtar
4710-057 Braga
Portugal



Copyright ©2026 ICVS. All Rights Reserved
Address
Life and Health Sciences
Research Institute (ICVS)
School of Medicine,
University of Minho,
Campus de Gualtar
4710-057 Braga
Portugal